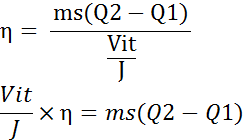Work Power & Energy
WORK
When force is applied on a body and that body covers certain distance in the direction of the force, the work is said to be done.
If
f = applied force
d = distance covered
w = work done
Then mathematically we can write
W = fd
the unit of work is joule in MKS system.
JOULE
If one Newton force is applied on a body and the covers a distance of one meter in the direction of force, then one joule of work is said to be done.
In fps system or British engineering system the unit is foot-pound.
FOOT-POUND (ft-lb)
When one-pound force is applied on a body and that body covers a distance of one foot in the direction of force, then one foot pound of work is said to be done.
1ft - 1b = 1.356 joule
1joule = 0.7375
POWER
The rate of doing work is called power. It is represented by "p" its unit is joule/sec or vs att.
WATT
When joule of work is done in one second then one-watt power is used. Power is the product of voltage and current of the electric circuit.
P=VI in BES, the unit of power is horsepower.
HORSE POWER
When 550 ft-lb of work is done in one second, then 1 horsepower of power is used.
1 hp = 550ft - lb/sec
= 550 x 1.356 joule/sec
= 550 x 1.356 watt
= 746 watt
ENERGY
The capability of a body to do work is called energy.. Mathematically energy is given as
Energy = Power x time
E = P x T
= vit joule
= iR * i * t joule
E = i2Rt joule
E = (V2/R)t joule
1 unit-=1 kilo watt hours = kwh
HEATING EFFECT OF CURRENT
Whenever current flows through a circuit, then the gets warmed and electrical energy is converted in to heat energy. We know that current is noting but the flow of electrons, so when electron enter in the conductor, they collide with the atoms and electron of it. As a result of these multiple collision heat is generated so this is the heating effect of the current.
JOULES LAW
When one-ampere current is flowing through a resistance, r ohm in time t second then the work done will be given as fallows.
W.d = vit joule
= i2Rt joule
= (V2/R)t joule
So electrical energy is converted in to heat energy. Heat energy is represented by "h". Heat energy is given by
work done
amount of heat energy = work done / mechenical equivalent of heat
H = WD / J
Where j=4200 joul/k.cal
KILO CALORIES (k.cal)
When one-kilo gram water is heated through one-degree cent grade. Then the amount of heat energy will be I k.cal.
THERMAL EFFICIENCY
Thermal efficiency is the rate between the useful heat energy actually used in heating the water and total heat energy generated.
heat actually utilized for heating the water
TE = heat actually utilized for heating the water / total heat produced electrically
heat actually utilized for heating the water =ms(Q2-Q1)
Where
m = mass of water in gram
S=time in second
Q1=initial temp of water
Q2=final temp of water
Heat produced electrically = vit/j
= I2Rt / J kwh
= V2t / RJ kwh
Thermal efficiency is represented by eta (η)